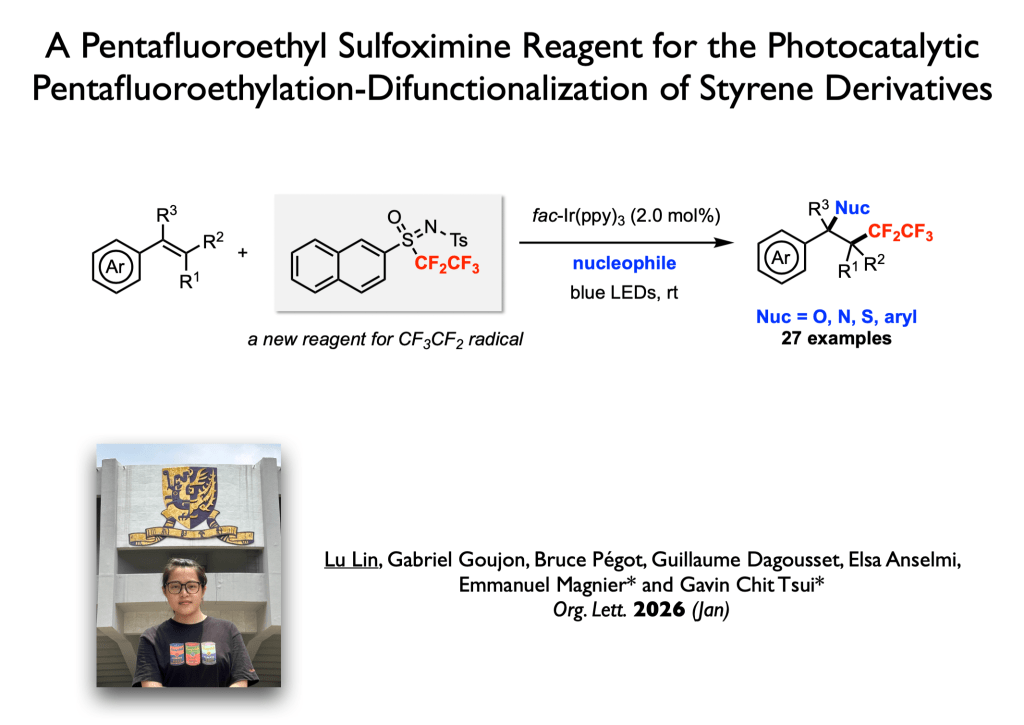
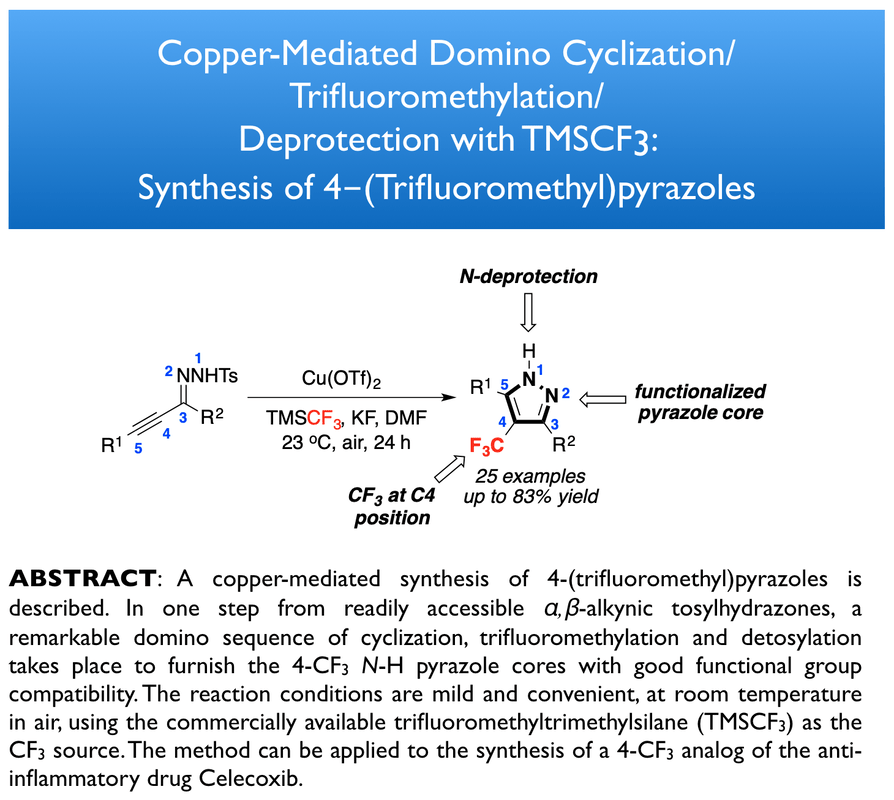
79. Lin, L.; Goujon, G.; Pegot, B.; Dagousset, G.; Anselmi, E.; Magnier, E.;* Tsui, G. C.* Org. Lett. 2026, 28, 1715-1719. (open access)
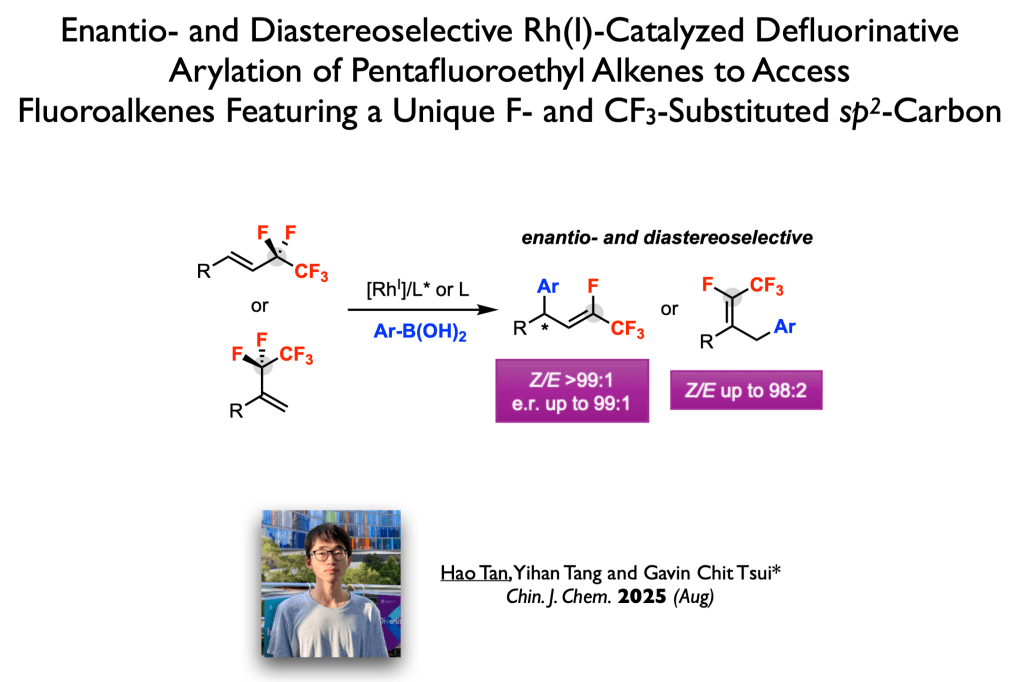
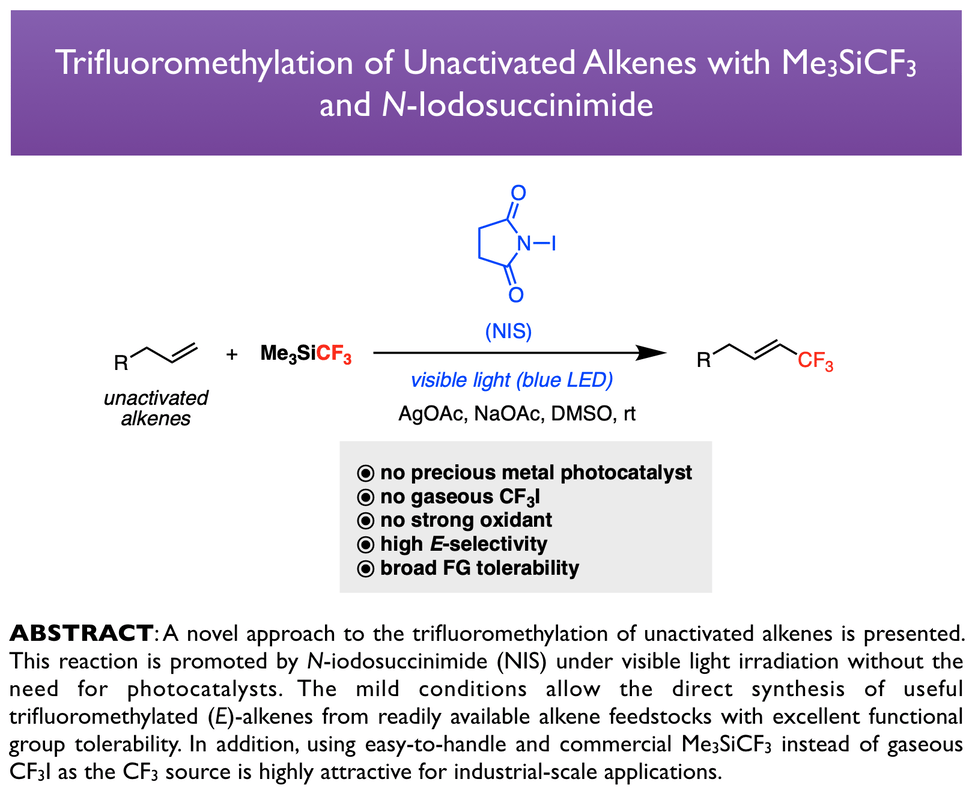
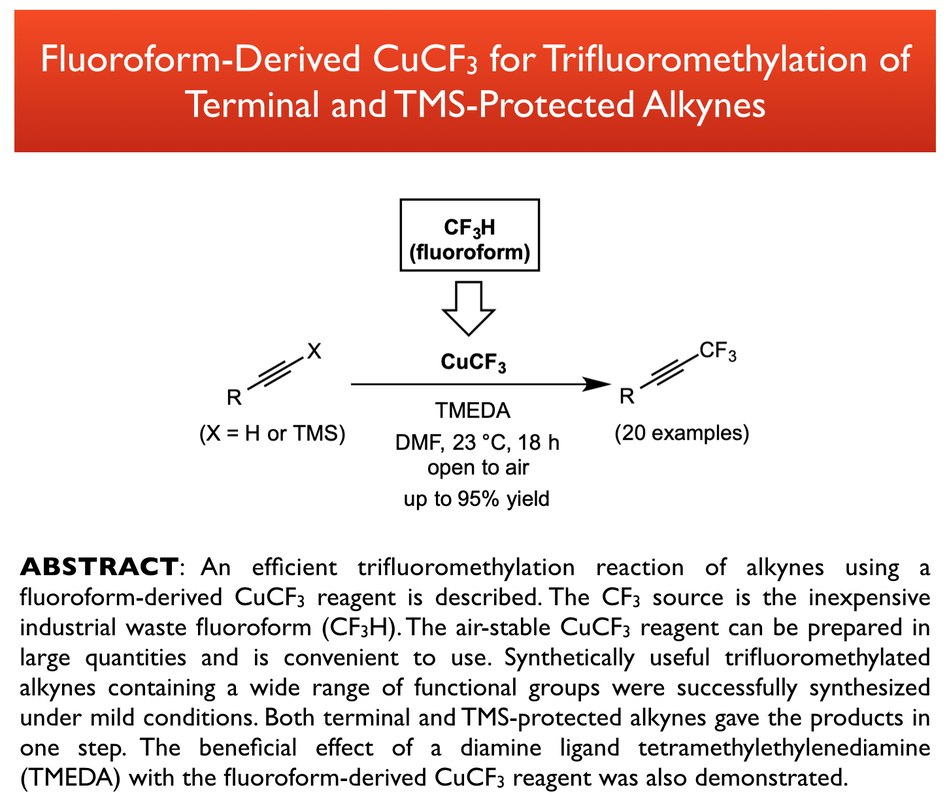
78. Tan, H.; Tang, Y.; Tsui, G. C.* Chin. J. Chem. 2025, 43, 3535-3542 (special issue of fluorine chemistry). (open access)
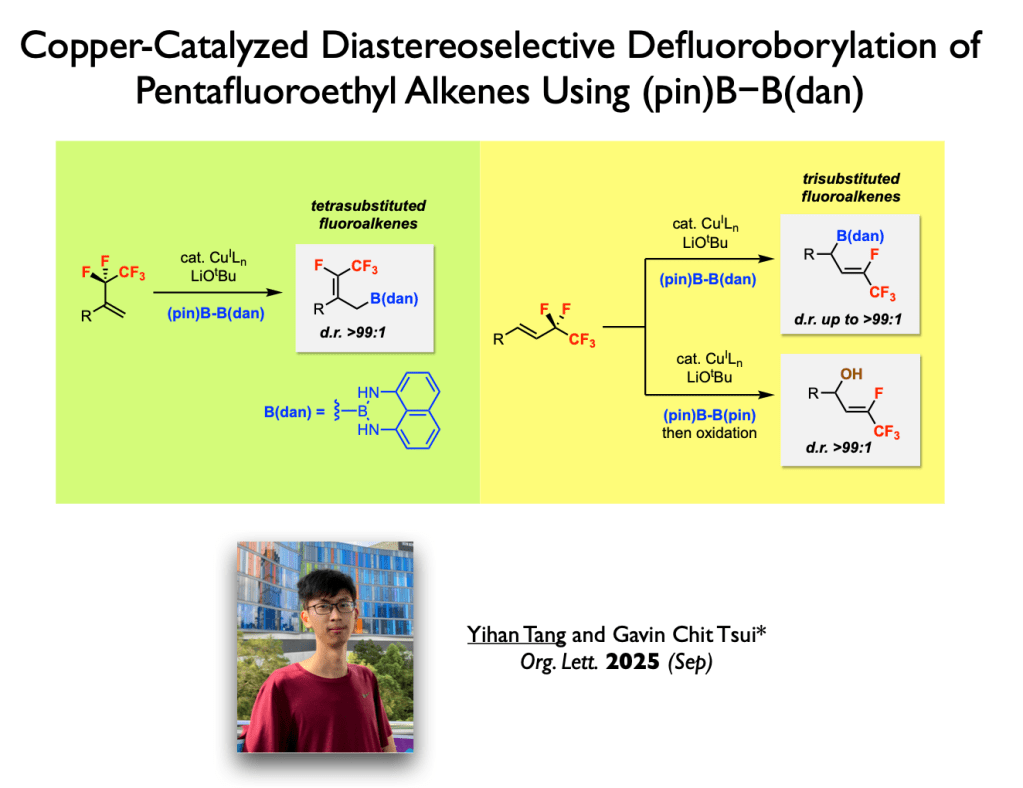
77. Tang, Y.; Tsui, G. C.* Org. Lett. 2025, 27, 11273-11277. (open access)
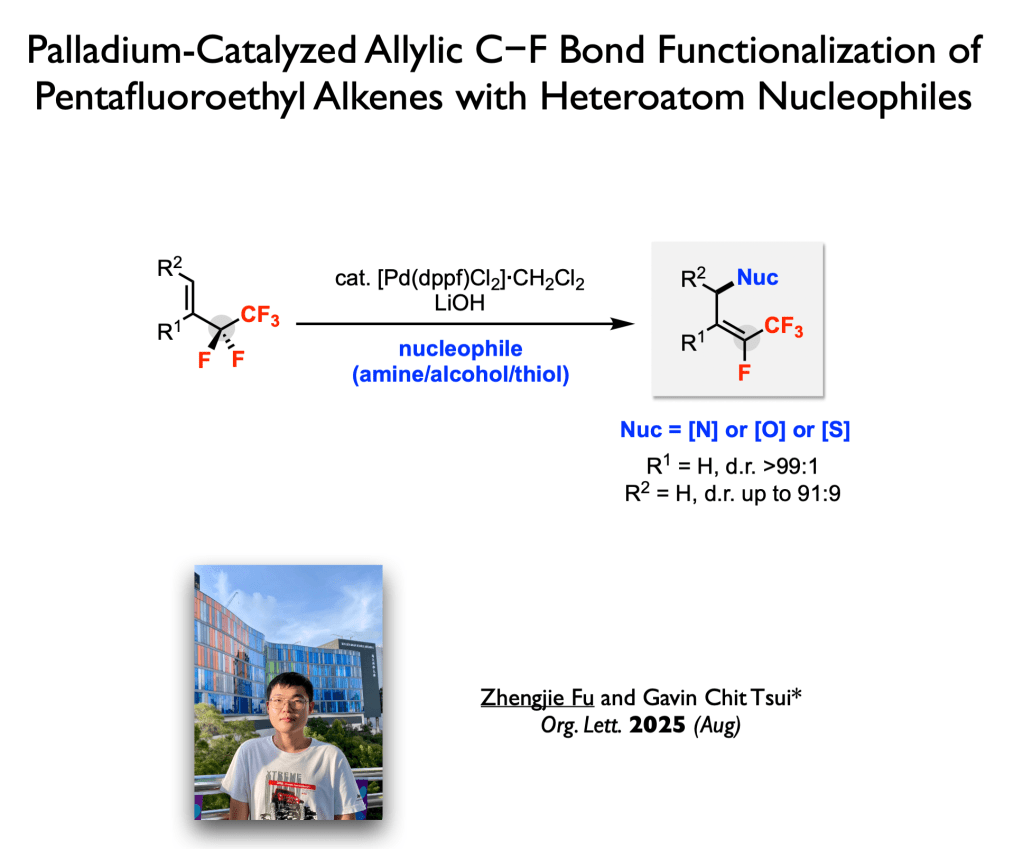
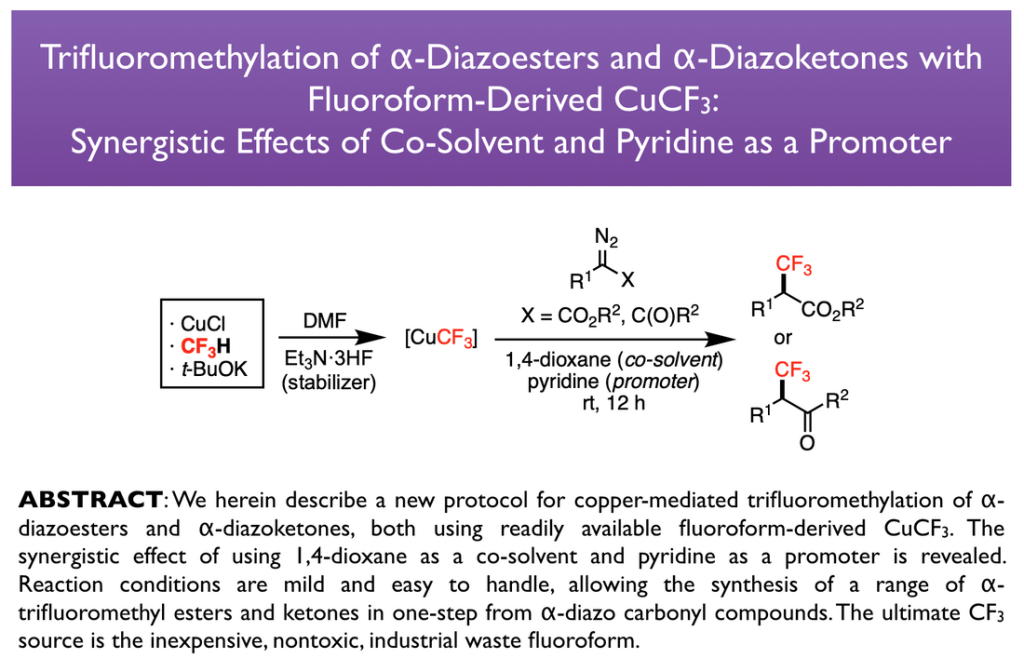
76. Fu, Z.; Tsui, G. C.* Org. Lett. 2025, 27, 10141-10146. (open access)
75. Zhao, Y.; Tsui, G. C.*; Shen, Q.* Chin. Chem. Lett. 2025, 36, 111051.
Key intermediates in Cu(I) to Cu(III) catalytic cycle for ethoxycarbonyl difluoromethylation
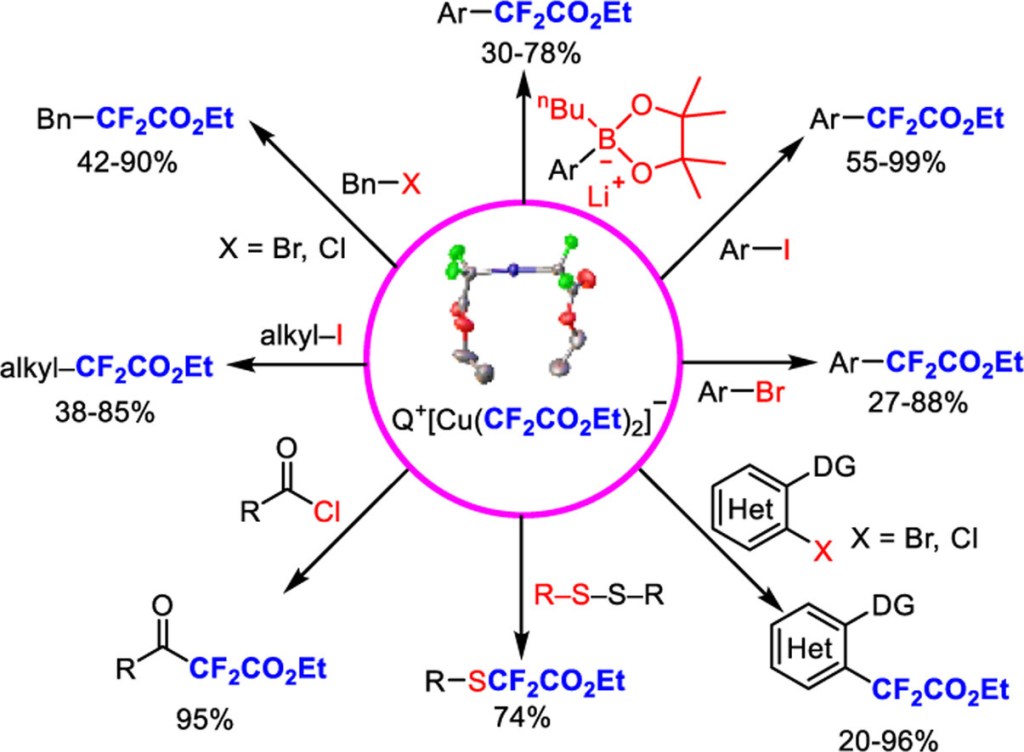
74. Tang, Y.; Tsui, G. C.* Org. Chem. Front. 2024, 11, 4366-4370.


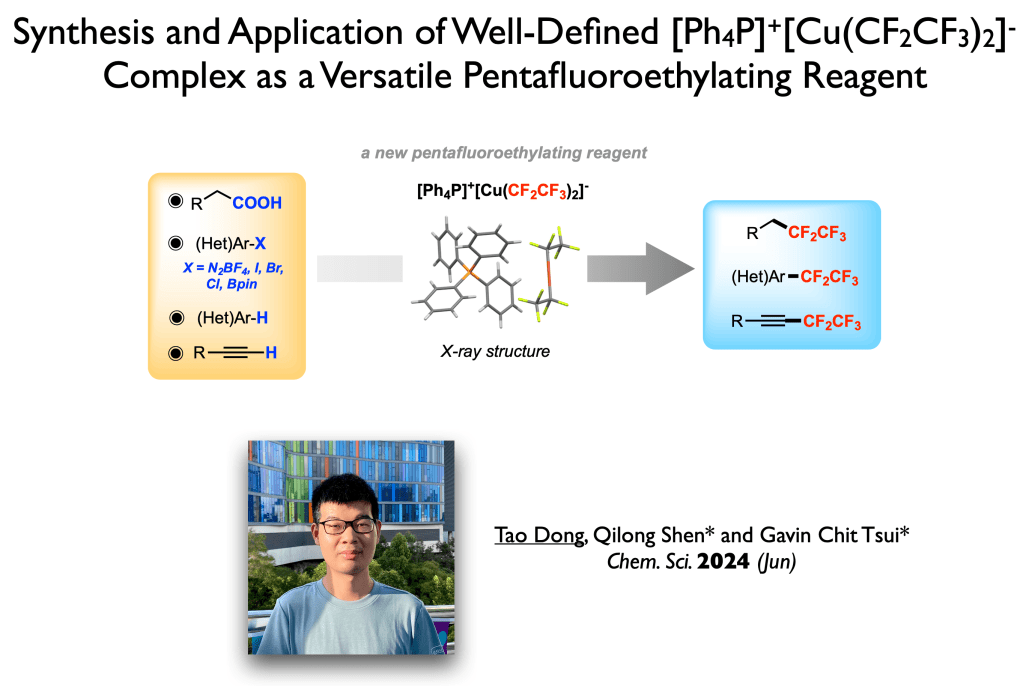
73. Dong, T.; Shen, Q.;* Tsui, G. C.* Chem. Sci. 2024, 15, 11550-11556.
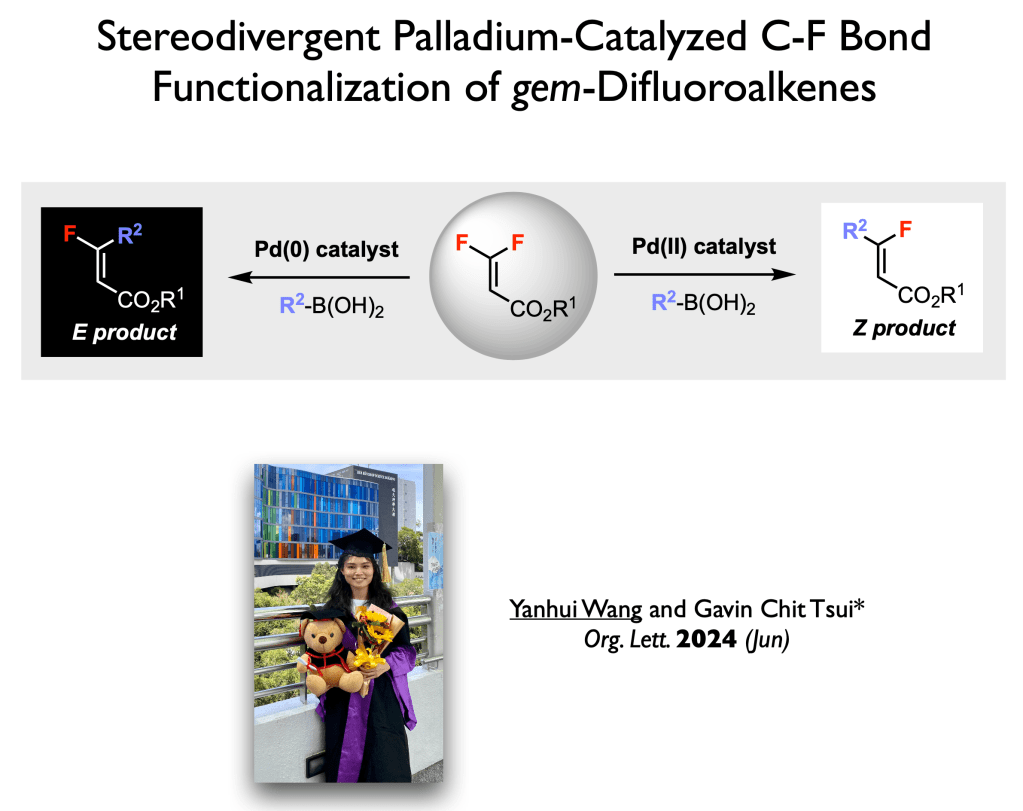
72. Wang, Y.; Tsui, G. C.* Org. Lett. 2024, 26, 5822-5826 (open access).
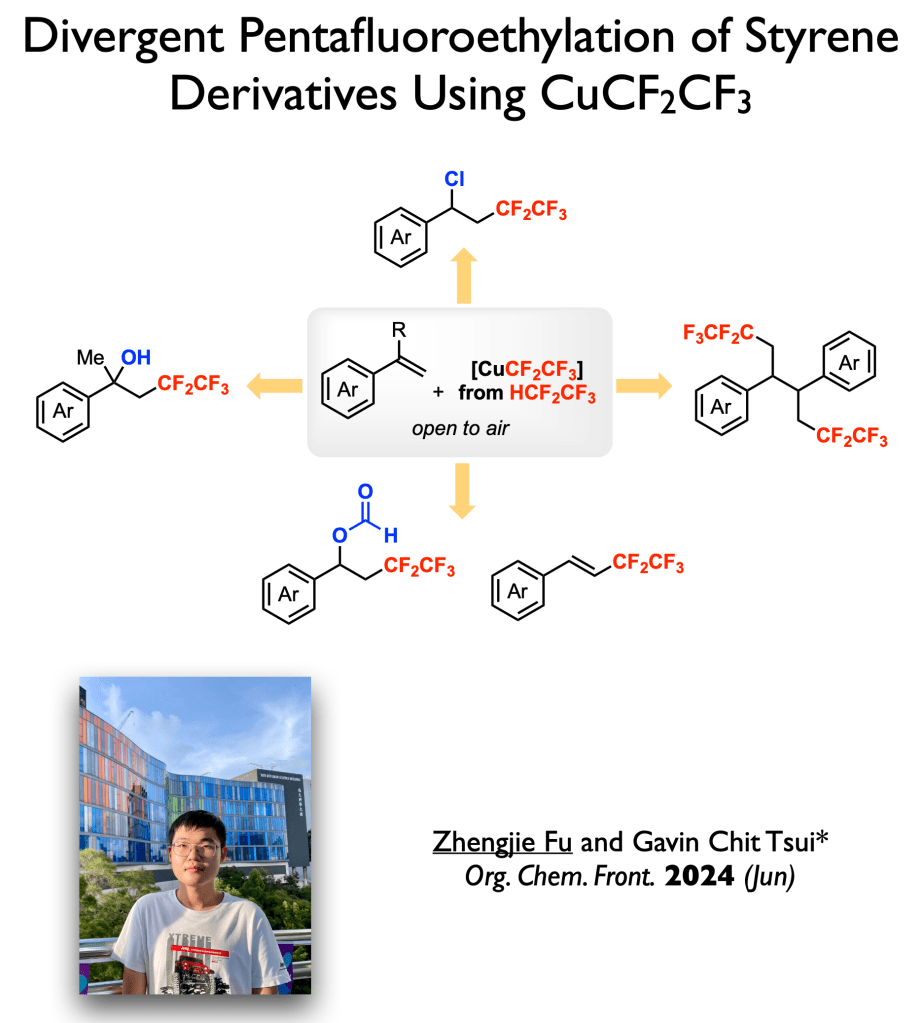
71. Fu, Z.; Tsui, G. C.* Org. Chem. Front. 2024, 11, 4697-4701.
70. Li, M.; Tsui, G. C.* Org. Lett. 2024, 26, 2223-2227 (open access).
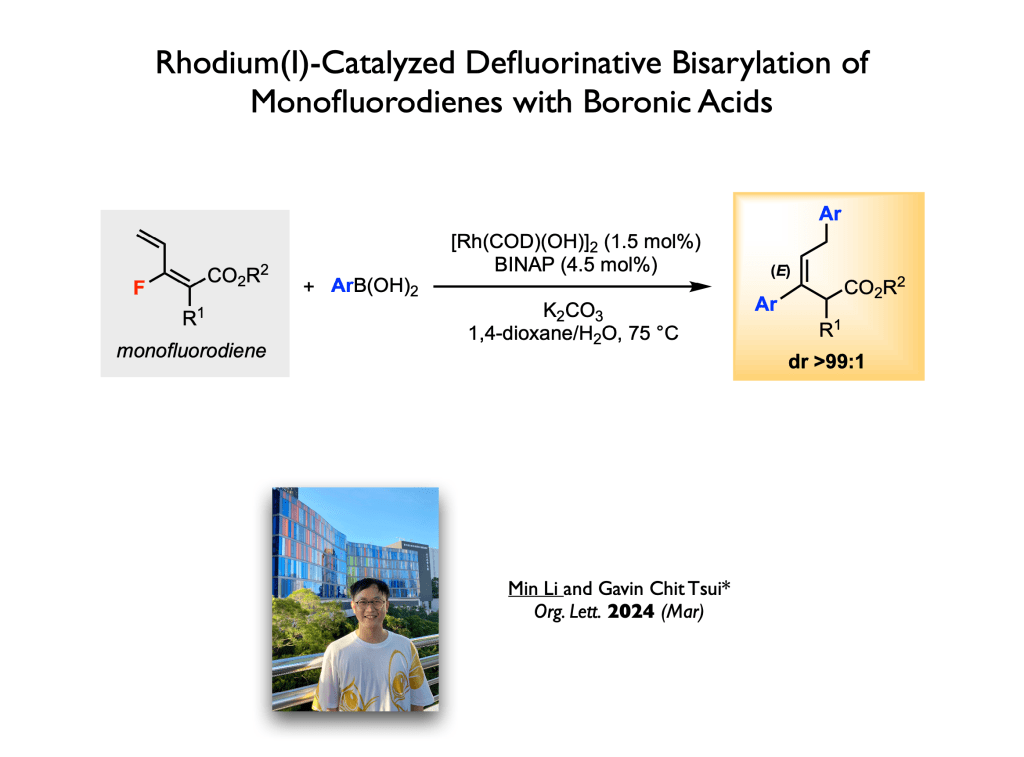
69. Zong, Y.; Tsui, G. C.* Org. Lett. 2024, 26, 1261-1264 (open access).
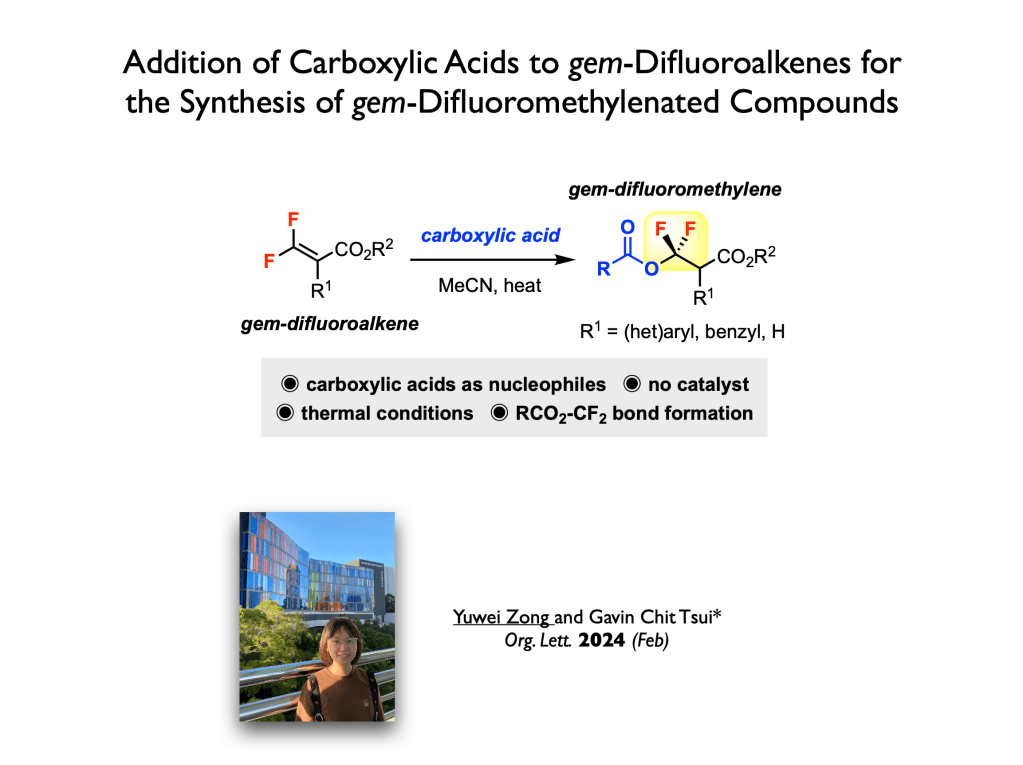
68. Li, M.; Tsui, G. C.* Org. Lett. 2024, 26, 376-379 (open access).
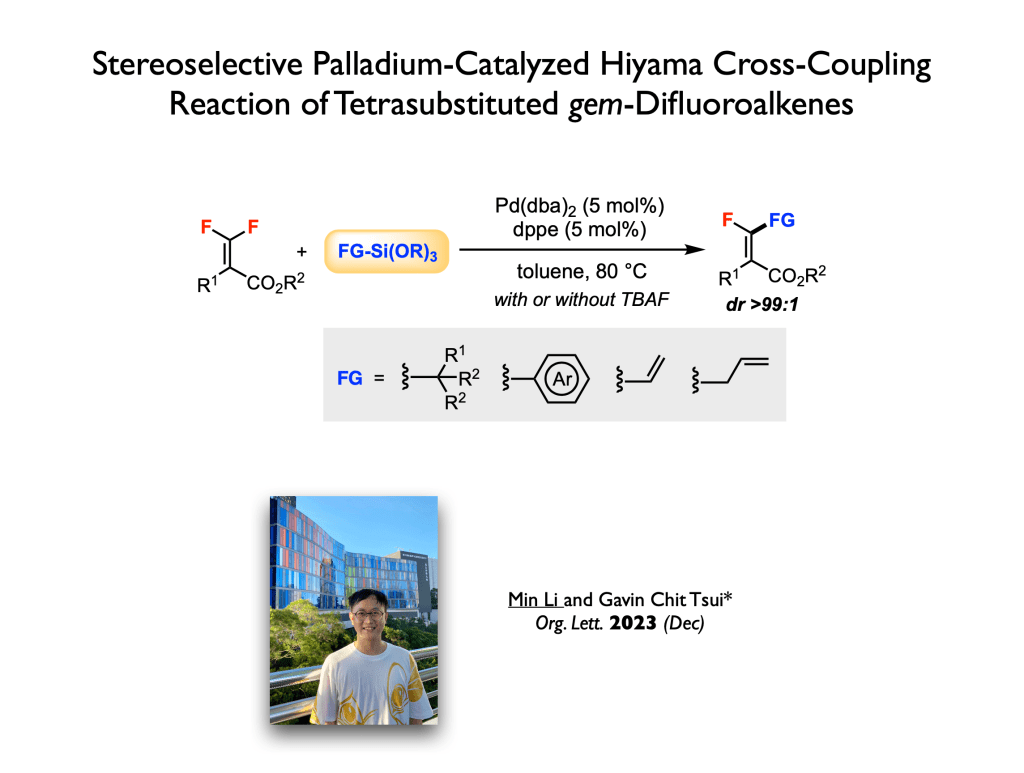
67. Tian, S.; Chen, M.; Tang, Y.; Cheng, K.;* Tsui, G. C.;* Wang, Q.* Org. Chem. Front. 2023, 10, 6124-6130.
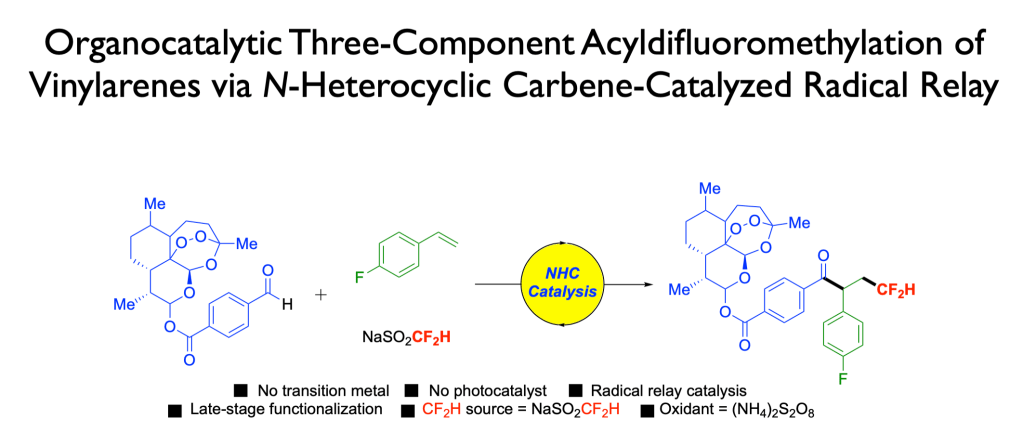
66. Dong, T.; Tang, Y.; Tsui, G. C.;* Org. Chem. Front. 2023, 10, 5092-5098. (highlight in Chinese)
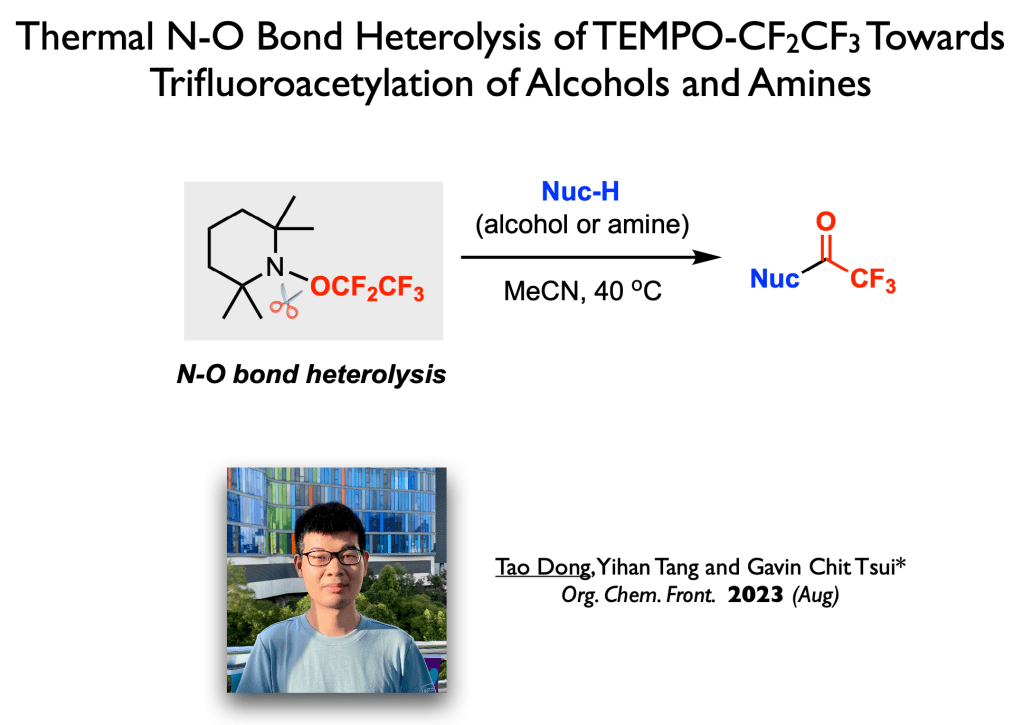
65. Dong, T.;+ Ye, Y.;+ Wang, Y.; Cheung, K. P. S.; Tsui, G. C.;* Chem. Asian J. 2023, 18, e202300655. (open access) +equal contribution – Celebrating the 60th Anniversary of The Chinese University of Hong Kong

64. Wang, Y; Tsui, G. C.;* Org. Lett. 2023, 25, 6217-6221. (open access)
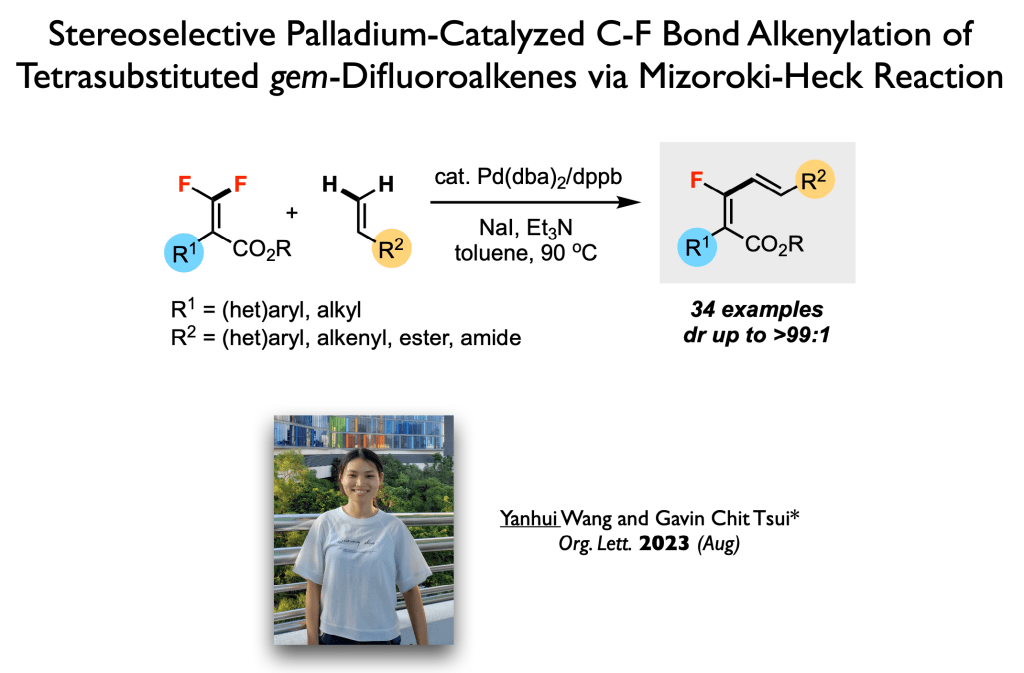
63. Luo, Z.; Chen, J.; Li, M.; Tsui, G. C.;* Org. Lett. 2023, 25, 5656-5660.
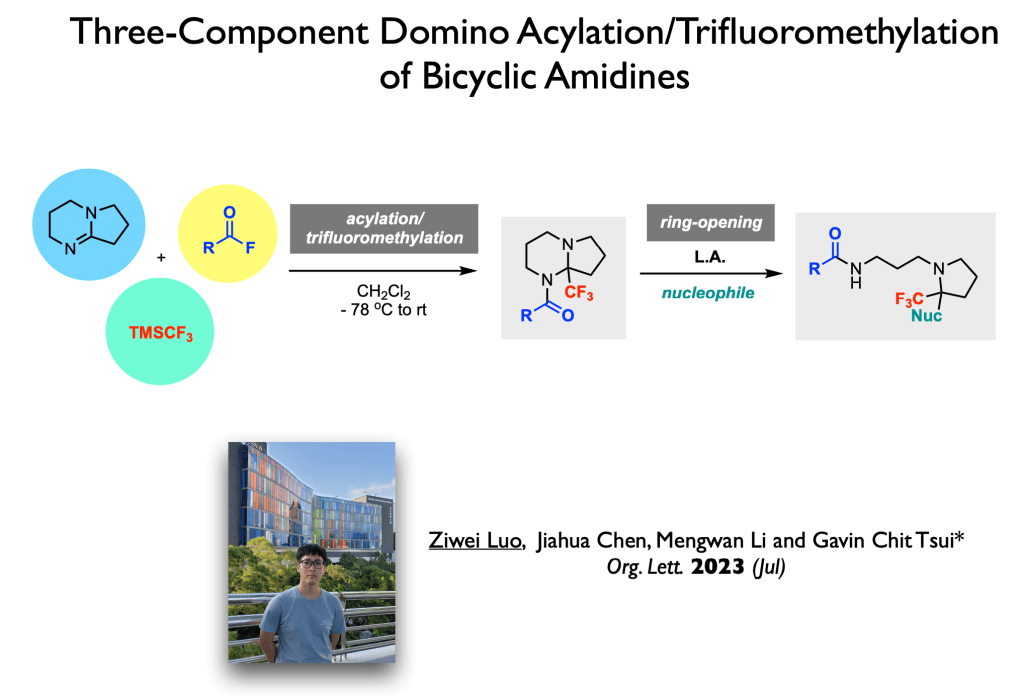
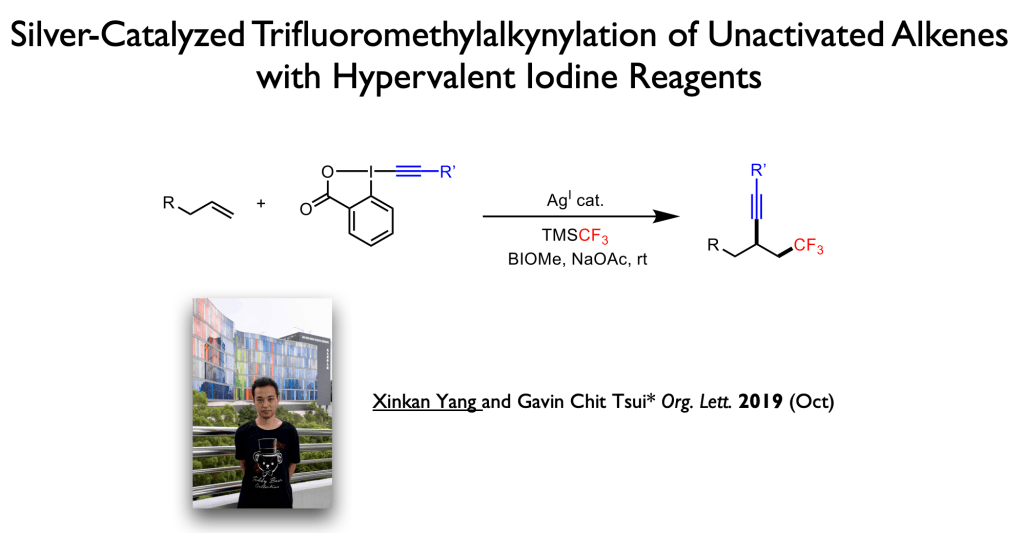
62. Fu, Z.; Yang, X.; Tsui, G. C.;* Org. Lett., 2023, 25, 4945-4949.
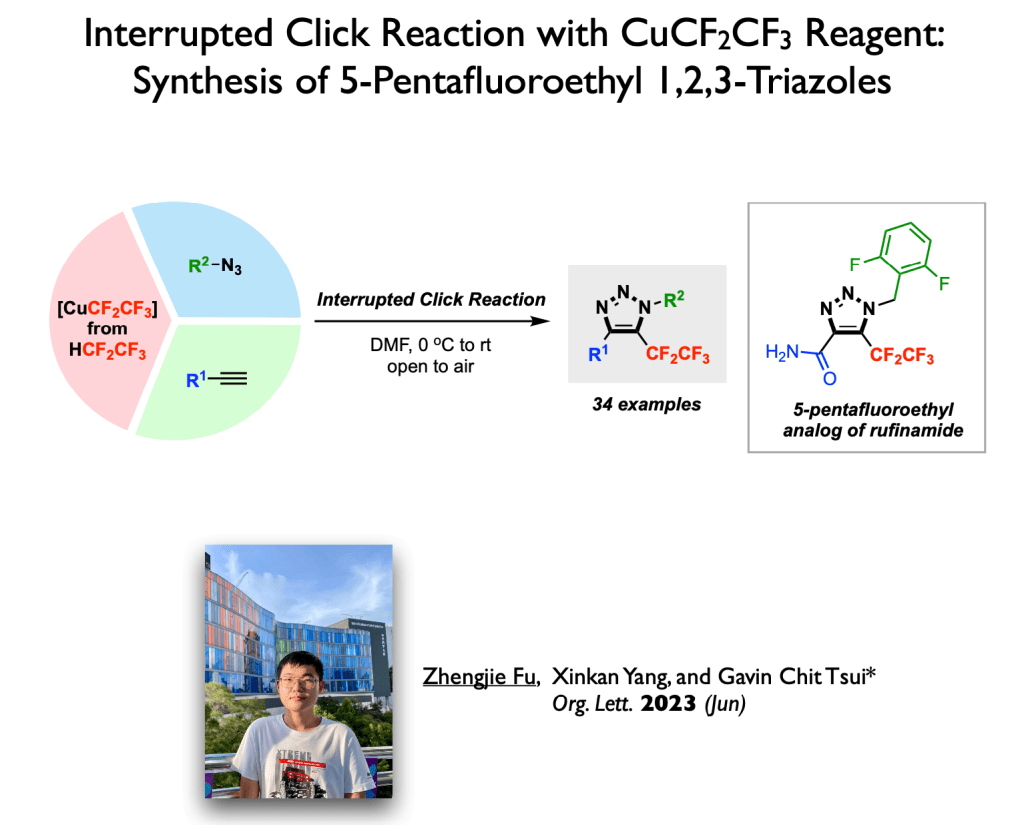
61. Luo, Z.; Zong, Y.; Tsui, G. C.;* Org. Lett., 2023, 25, 4406-4410.
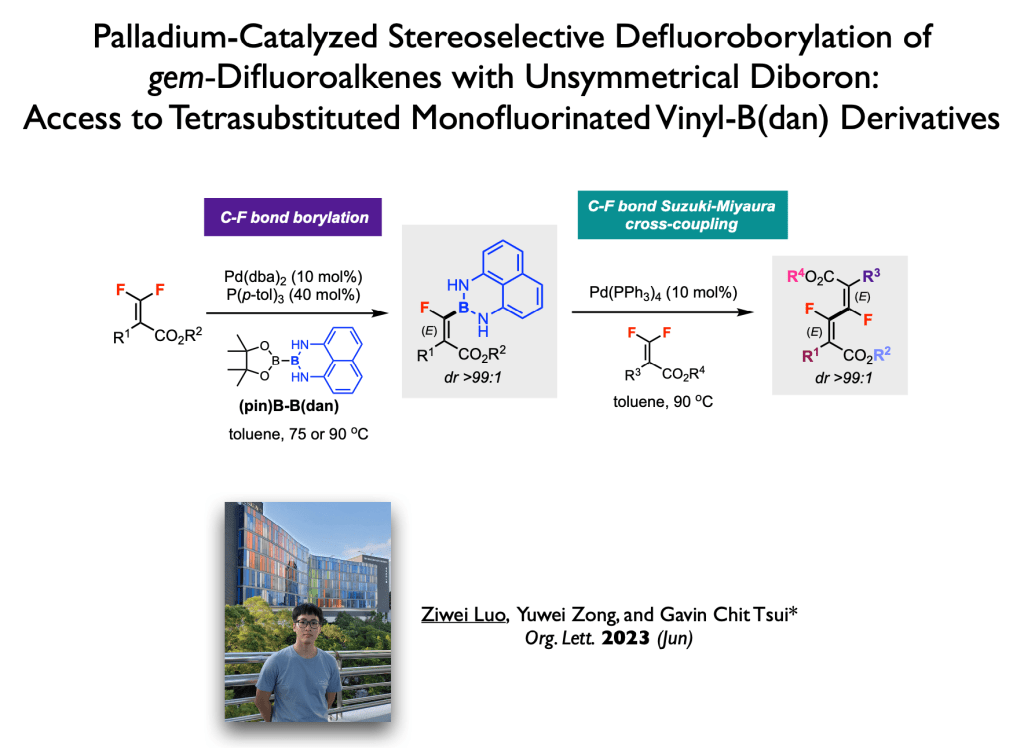
60. Zong, Y.; Luo, Z.; Tsui, G. C.;* Org. Lett., 2023, 25, 2333-2337.

59. Lin, L.; Wang, P.; Dong, T.; Tsui, G. C.;* Liao, S.* Org. Lett., 2023, 25, 1088-1093.
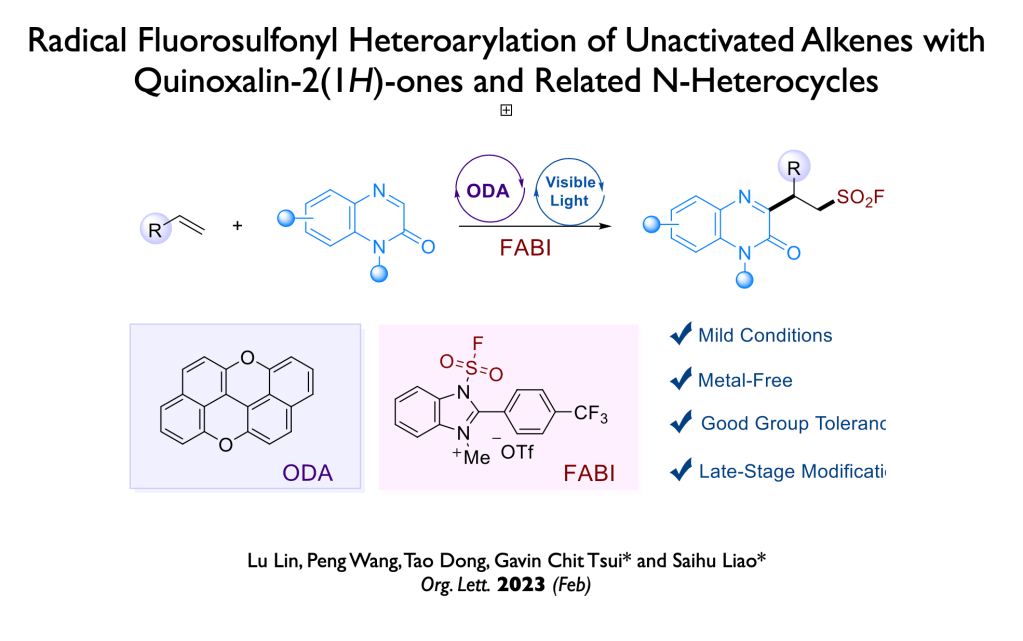
58. Wang, J.; Luo, Z.; Wu, Y.; Tang, Y.; Yang, X.;* Tsui, G. C.;* Org. Lett., 2023, 25, 1045-1049.

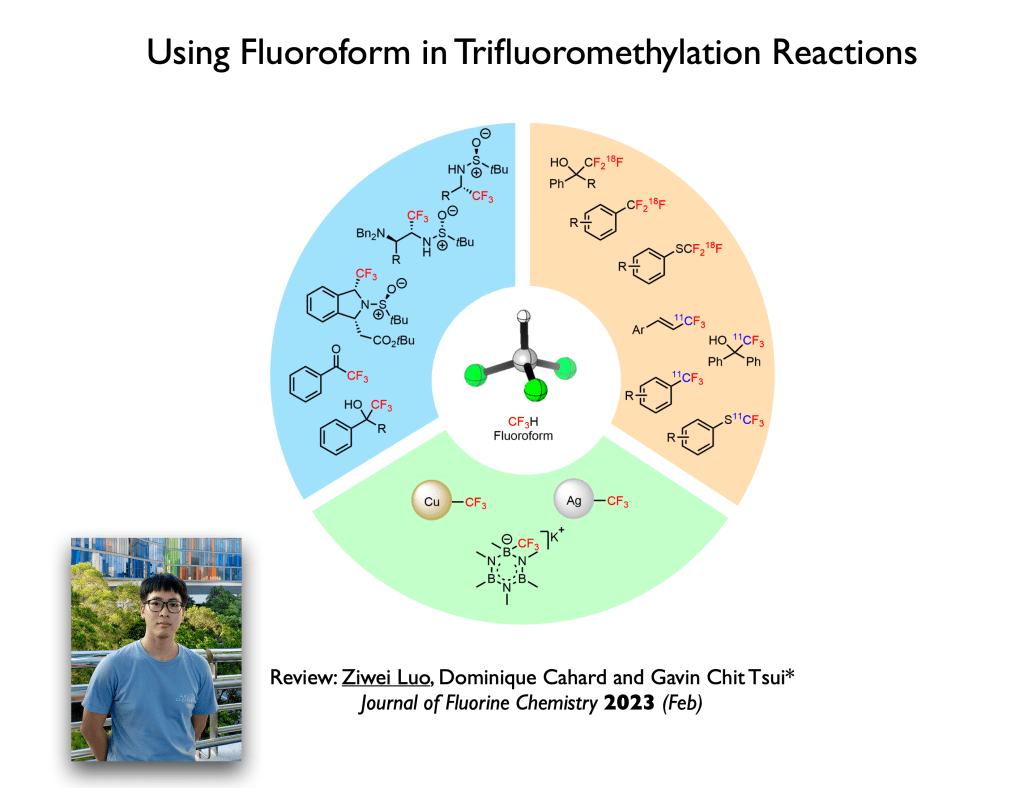
57. Review: Luo, Z.; Cahard, D.;* Tsui, G. C.;* Journal of Fluorine Chemistry, 2023, 266, 110092.
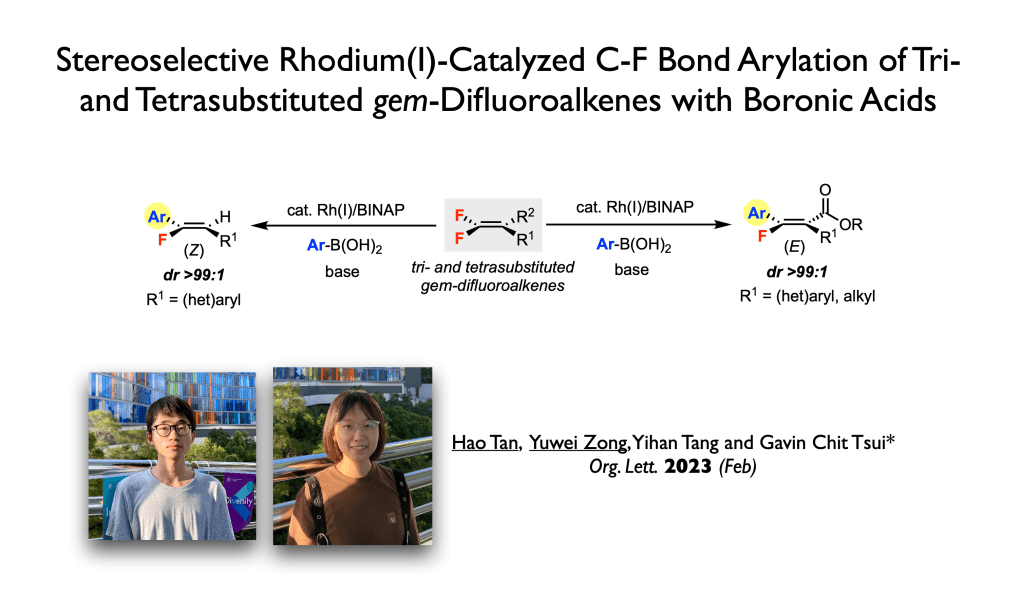
56. Tan, H.; Zong, Y.; Tang, Y.; Tsui, G. C.;* Org. Lett. 2023, 25, 877-882.
55. Zong, Y.; Tang, Y.; Tsui, G. C.;* Org. Lett. 2022, 24, 6380-6385. (Front cover)


54. Luo, Z.; Tsui, G. C.;* Org. Chem. Front. 2022, 9, 4969-4974.
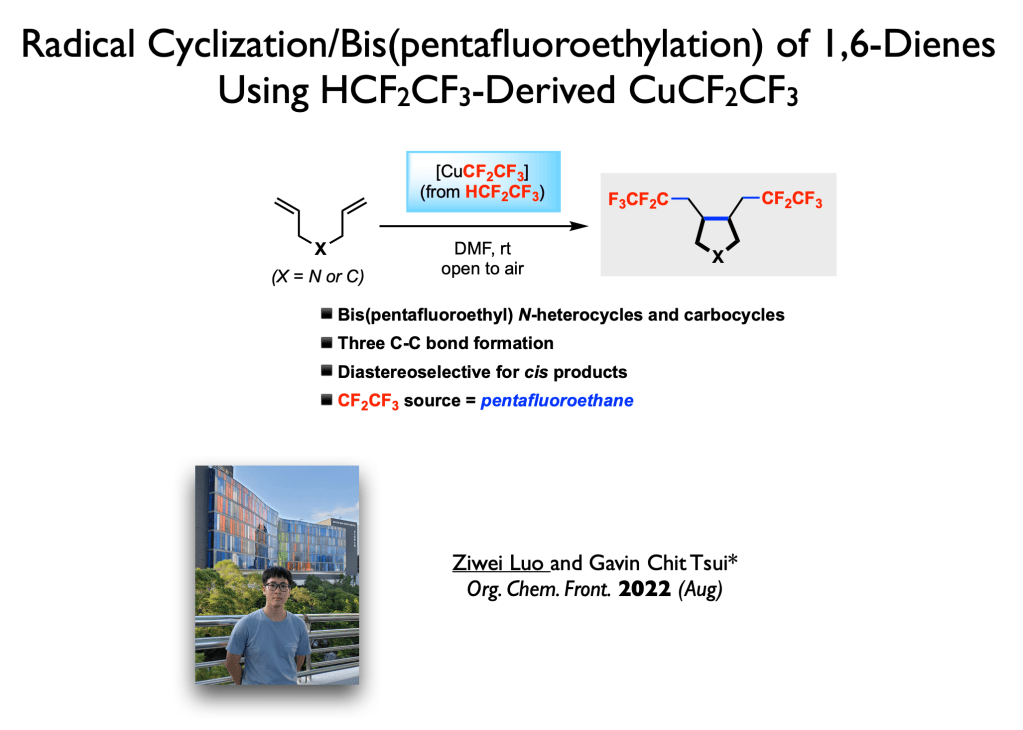
53. She, K; Liang, F.; Tian, S.; Wang, H.;* Tsui, G. C.;* Wang, Q.* Org. Lett. 2022, 24, 4840-4844.
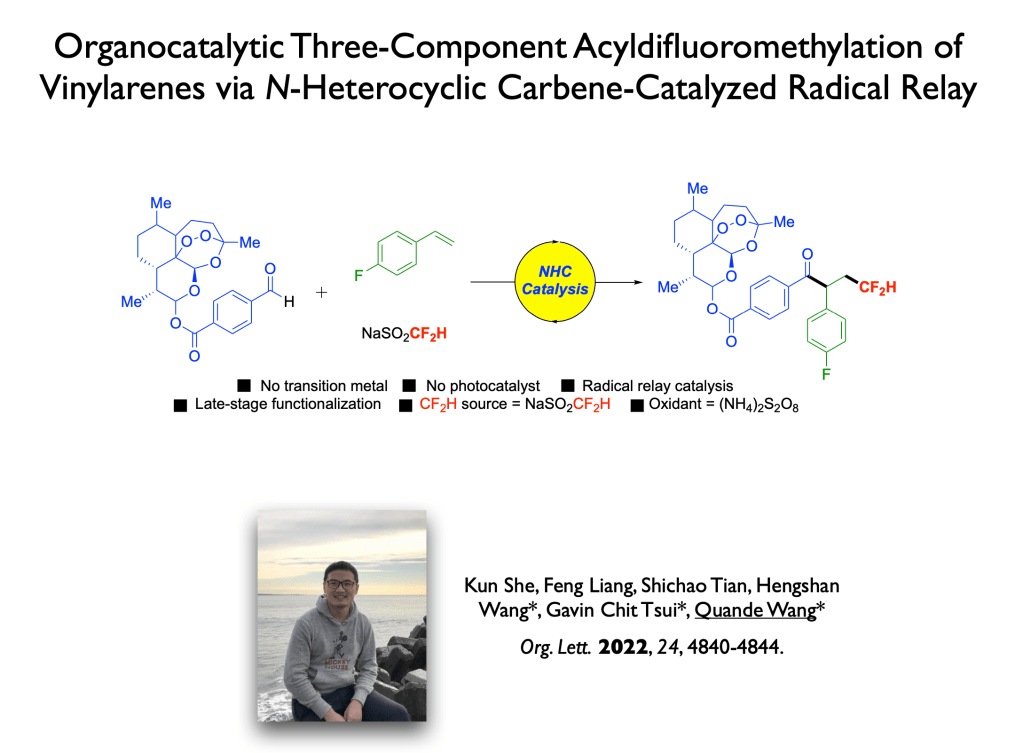
52. Liang, R.-X.,* Chen, J.-F.; Huang, Y.-Y.; Yu, Y.-P.; Zhang, H.-Y.; Song, Y.-F.; Tsui, G. C.* Jia, Y.-X. Chem. Commun. 2022, 58, 6200-6203.
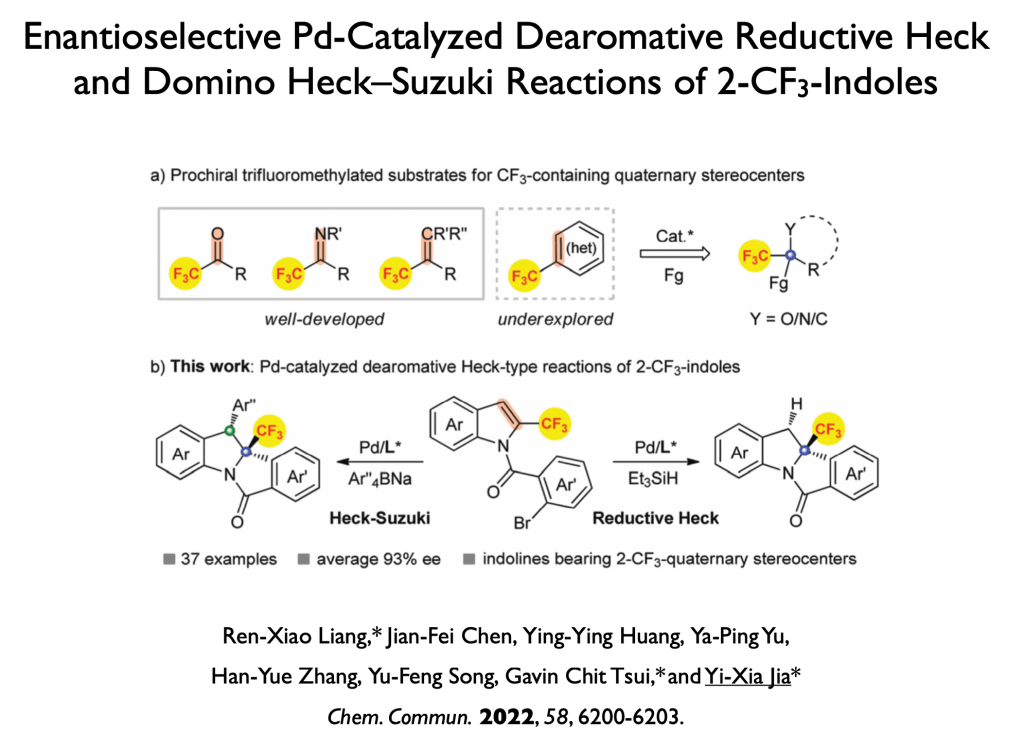
51. Wang, Y; Tang, Y.; Zong, Y.; Tsui, G. C.* Org. Lett. 2022, 24, 4087-4092.
50. Personal Account: Yang, X.; Tsui, G. C.* Synlett (Synpacts) 2022, 33, 713-720.
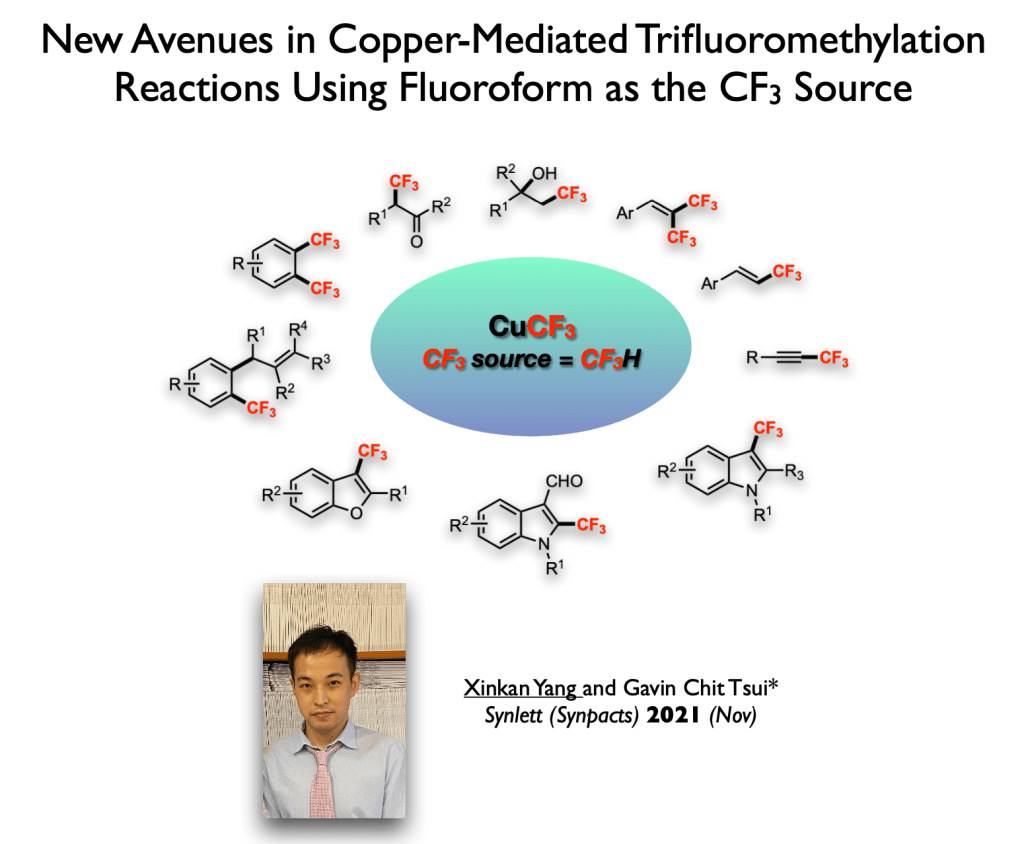
49. Review: Dong, T.; Tsui, G. C.* Chem. Rec. 2021, 21, 4015-4031.

48. Li, M.; Wang, Y.; Tsui, G. C.* Org. Lett. 2021, 23, 8072-8076.

47. Zong, Y.; Ma, Q.; Tsui, G. C.* Org. Lett. 2021, 23, 6169-6173.

46. Wang, Y.; Ma, Q.; Tsui, G. C.* Org. Lett. 2021, 23, 5241-5245.
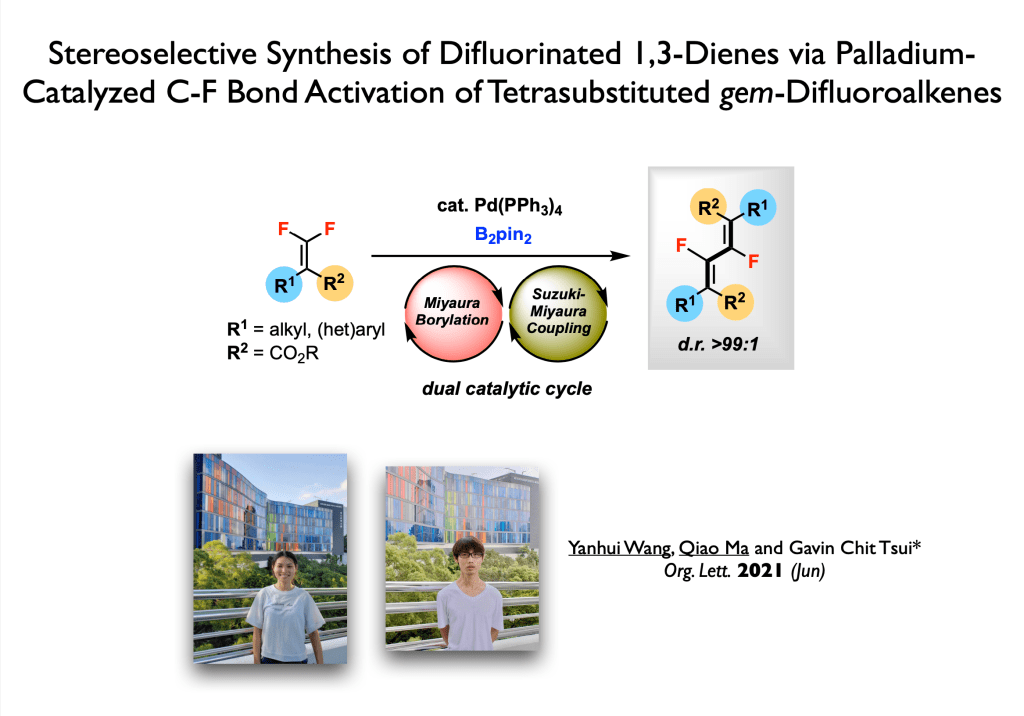
45. Wang, Y.; Qi, X.; Ma, Q.; Liu, P.;* Tsui, G. C.* ACS Catal. 2021, 11, 4799-4809.

44. Luo, Z.; Yang, X.; Tsui, G. C.* Org. Lett. 2020, 22, 6155-6159. Highlighted by ChemistryViews.

43. Ma, Q.; Liu, C.; Tsui, G. C.* Org. Lett. 2020, 22, 5193-5197.
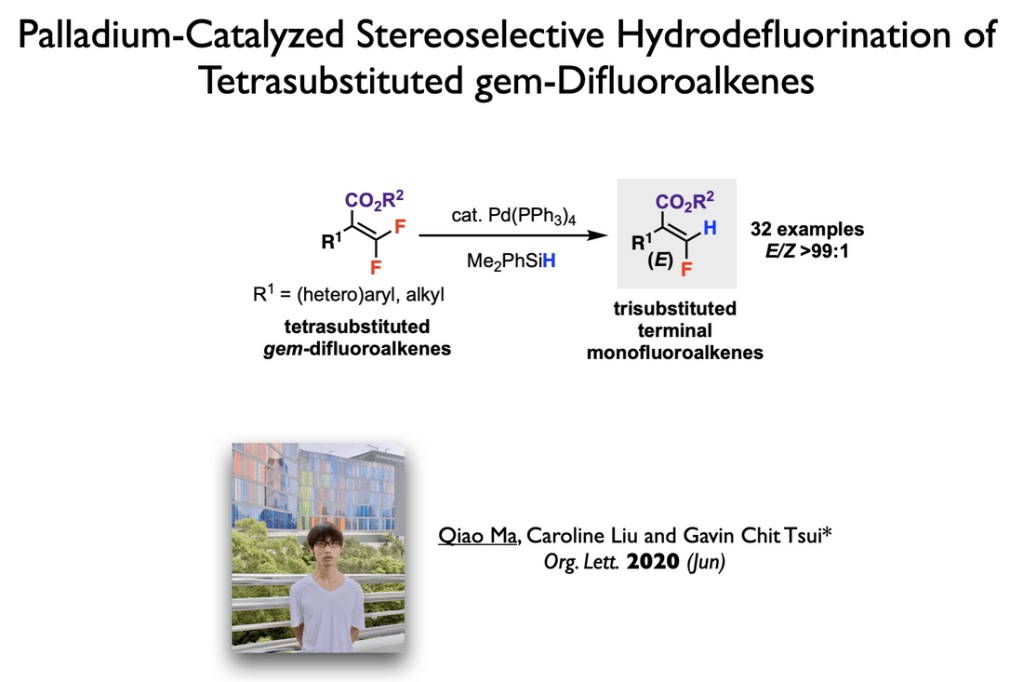
42. Yang, X.; Tsui, G. C.* Org. Lett. 2020, 22, 4562-4567.

41. Ma, Q.; Wang, Y.; Tsui, G. C.* Angew. Chem. Int. Ed. 2020, 59, 11293-11297. Highlighted by Synfacts 2020, 16(07), 0800.
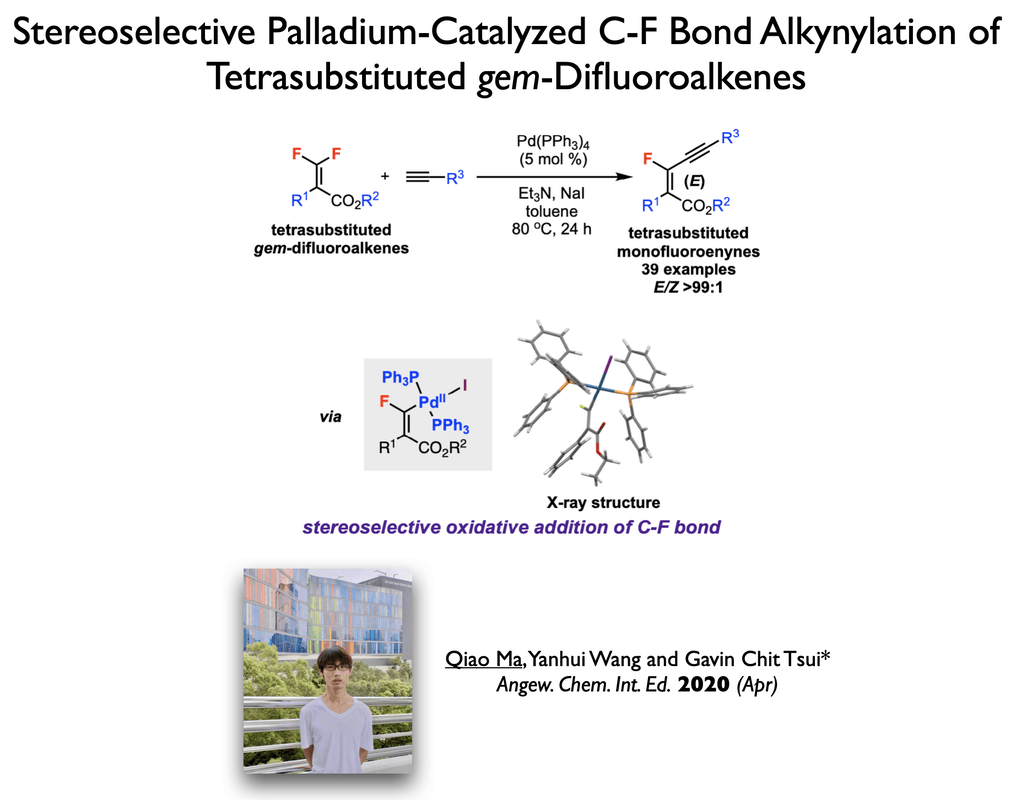
40. Yang, X.; Tsui, G. C.* Org. Lett. 2019, 21, 8625-8629.
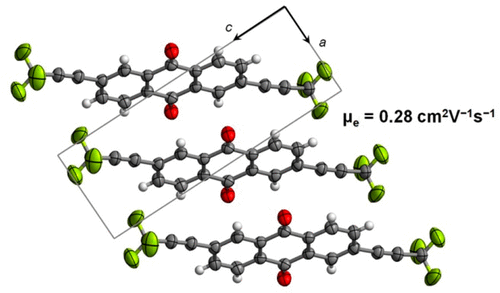
39. Trifluoromethylation of Anthraquinones for n-Type Organic Semiconductors in Field Effect Transistors. Zhao, M.; Yang, X.; Tsui, G. C.;* Miao, Q.* J. Org. Chem. 2020, 85, 44-51. (collaboration with Prof. Qian Miao, CUHK)
Special! Read our guest editorial on a special issue of Organofluorine Chemistry in the Asian Journal of Organic Chemistry (2019-May) Asian J. Org. Chem. 2019, 8, 566-567.

38. Yang, X.; Tsui, G. C.* Org. Lett. 2019, 21, 1521-1525 (one of the most downloaded papers in Feb 2019). See ChemistryViews for a highlight.
37. Li, M.; Ye, Y.; He, L.; Hui, M.; Ng, T. B.; Wong, J. H.; Tsui, G. C.* Asian J. Org. Chem. 2019, 8, 702-709 (special issue).

36. Ma, Q.; Tsui, G. C.* Org. Chem. Front. 2019, 6, 27-31.
35. Yang, X.; Tsui, G. C.* Chem. Sci. 2018, 9, 8871-8875. Highlighted by Synfacts 2018, 14(12), 1291.

34. Ye, Y.; Cheung, K. P. S.; He, L.; Tsui, G. C.* Org. Chem. Front. 2018, 5, 1511-1515.
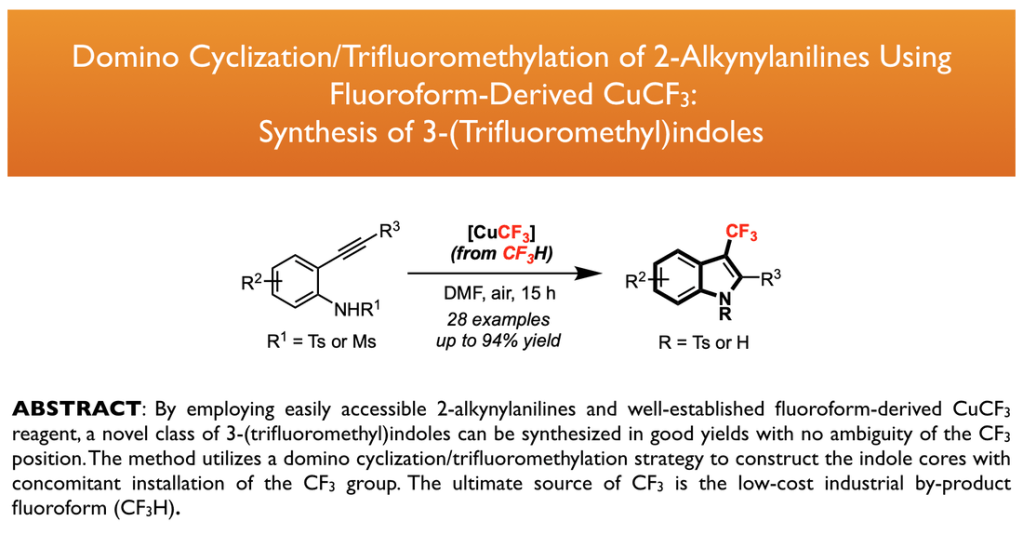
33. Ye, Y.; Cheung, K. P. S.; He, L.; Tsui, G. C.* Org. Lett. 2018, 20, 1676-1679. Highlighted by Synfacts 2018, 14(05), 0459. Highlighted by Organic Chemistry Portal.

32. Wang, Q.; Tsui, G. C.* J. Org. Chem. 2018, 83, 2971-2979.
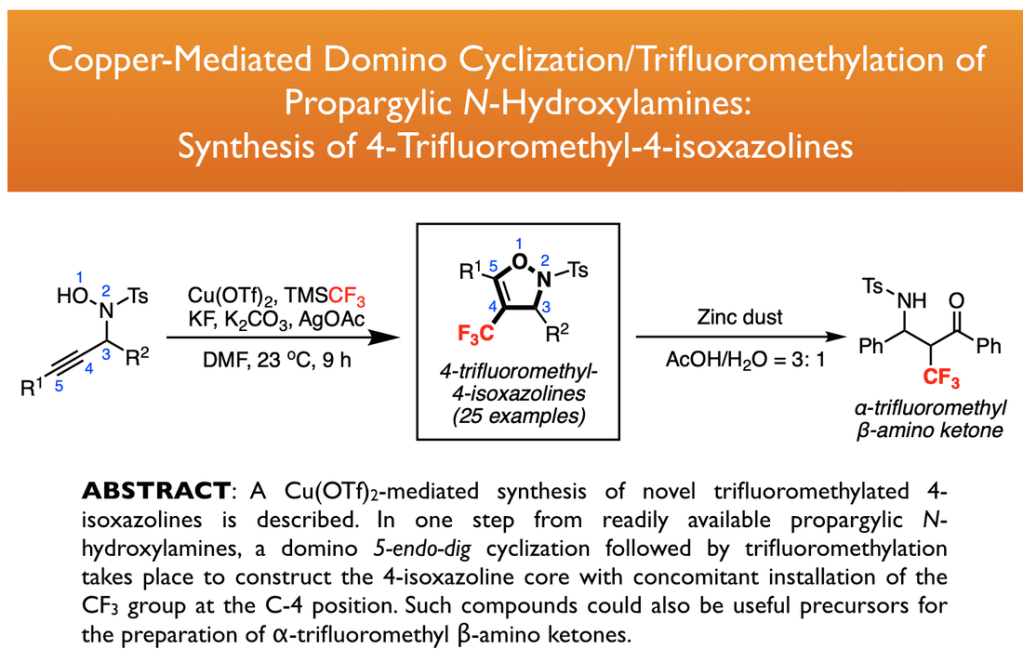
31. Yang, X.; Tsui, G. C.* Org. Lett. 2018, 20, 1179-1182.
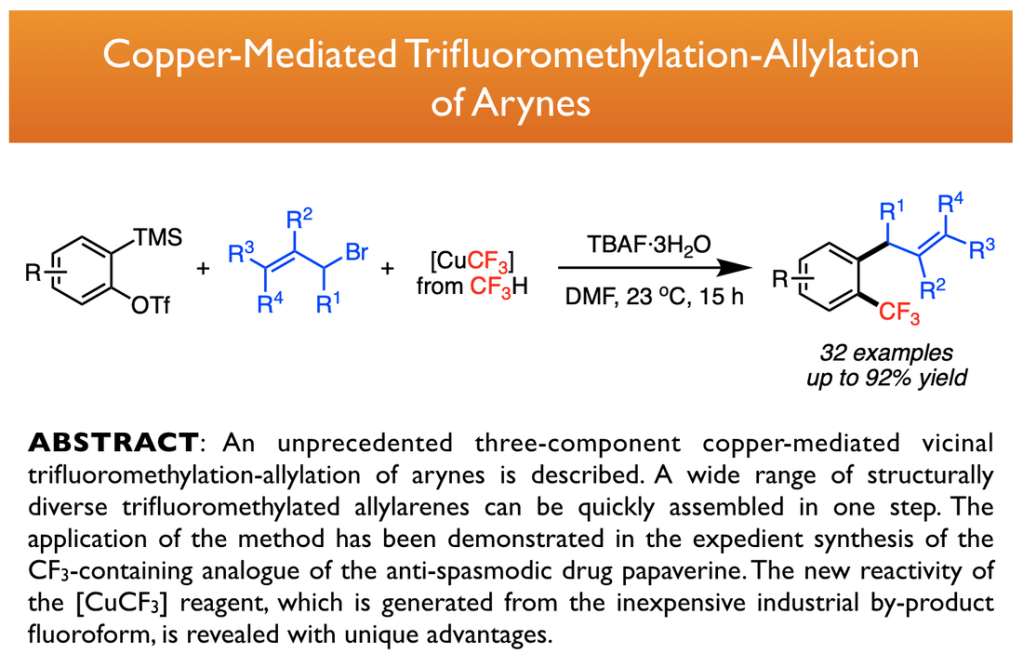
30. He, L.; Yang, X.; Tsui, G. C.* J. Org. Chem. 2017, 82, 6192-6201. Highlighted by Synfacts 2017, 13(08), 0857. Highlighted by Organic Chemistry Portal.

29. Cheung, K. P. S.; Tsui, G. C.* Org. Lett. 2017, 19, 2881-2884.

28. Yang, X.; He, L.; Tsui, G. C.* Org. Lett. 2017, 19, 2446-2449. Highlighted by Synfacts 2017, 13(07), 0749.

27. Synthesis of Fluorescent Indazoles by Palladium-Catalyzed Benzannulation of Pyrazoles with Alkynes. Kim, O. S.; Jang, J. H.; Kim, H. T.; Han, S. J.; Tsui, G. C.*; Joo, J. M.* Org. Lett. 2017, 19, 1450-1453. (collaboration with Prof. Jung Min Joo, PNU, South Korea)
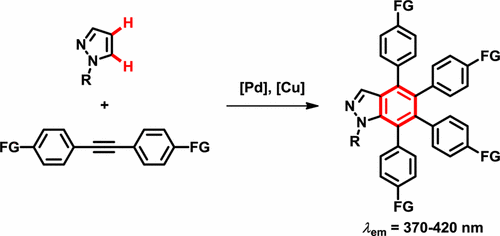
26. Wang, Q.; He, L.; Li, K. K.; Tsui, G. C.* Org. Lett. 2017, 19, 658-661. Highlighted by Organic Chemistry Portal.
25. Review: Fluoroform, First Update. He, L.; Tsui, G. C.*, In Encyclopedia of Reagents for Organic Synthesis, Rovis, T. Ed.; Wiley-VCH: Weinheim, 2017.

24. Book chapter: Five-Membered Ring Systems: Furans and Benzofurans. Ke, Z.; Tsui, G. C., Peng, X.-S.; Yeung, Y.-Y., In Progress in Heterocyclic Chemistry, Gribble, G. W.; Joule, J. A. Eds.; Elsevier: Oxford, 2016 to 2020.

23. He, L.; Tsui, G. C.* Org. Lett. 2016, 18, 2800-2803. Highlighted by Synfacts 2016, 12(08), 0840. Highlighted by Organic Chemistry Portal.
Before Independent Career
22. The Organocatalytic Asymmetric Prins Cyclization. Tsui, G. C.; Liu, L.; List, B. Angew. Chem. Int. Ed. 2015, 54, 7703-7706.
21. Review: CH-Activation Reactions in Domino Processes. Tsui, G. C.; Lautens, M. In Domino Reactions – Concept for Efficient Organic Synthesis, Tietze, L. F. Ed.; Wiley-VCH: Weinheim, 2014.
20. Ruthenium-Catalyzed [2+2] Cycloadditions between Norbornene and Propargylic Alcohols or Their Derivatives. Tsui, G. C.; Villeneuve, K.; Carlson, E.; Tam, W. Organometallics, 2014, 33, 3847-3856.
19. Metal–Ligand Binding Affinity vs Reactivity: Qualitative Studies in Rh(I)-Catalyzed Asymmetric Ring-Opening Reactions. Tsui, G. C.; Dougan, P.; Lautens, M. Org. Lett. 2013, 15, 2652-2655.
18. Expedient Synthesis of Chiral Oxazolidinone Scaffolds via Rhodium-Catalyzed Asymmetric Ring-Opening Reaction with Sodium Cyanate. Tsui, G. C.; Ninnemann, N. M.; Hosotani, A.; Lautens, M. Org. Lett. 2013,15, 1064-1067.
17. Rhodium-Catalyzed Enantioselective Nucleophilic Fluorination: Ring Opening of Oxabicyclic Alkenes. Zhu, J.†; Tsui, G. C.†; Lautens, M. Angew. Chem. Int. Ed. 2012, 51, 12353-12356. († Equal contributions)
16. One-Pot Synthesis of Chiral Dihydrobenzofuran Framework via Rh/Pd Catalysis. Tsui, G. C.; Tsoung, J.; Dougan, P.; Lautens, M. Org. Lett. 2012, 14, 5542-5545.
15. Rhodium(I)-Catalyzed Domino Asymmetric Ring Opening/Enantioselective Isomerization of Oxabicyclic Alkenes with Water. Tsui, G. C.; Lautens, M. Angew. Chem. Int. Ed. 2012, 51, 5400-5404.
14. Use of (Z)-β-(ortho-Fluorobenzenesulfonyl)vinylamines as Novel Synthons in the Synthesis of 1,4-Benzothiazine Derivatives. Tsui, G. C.; Singjunla, Y.; Lautens, M. Synthesis 2012, 44, 1359-1364.
13. Asymmetric Synthesis of (Triaryl)methylamines by Rhodium-Catalyzed Addition of Arylboroxines to CyclicN-Sulfonyl Ketimines. Nishimura, T.; Noishiki, A.; Tsui, G. C.; Hayashi, T. J. Am. Chem. Soc. 2012, 134, 5056-5059.
12. 3,4-Diarylpiperidines as potent renin inhibitors. Lacombe, P.; Arbour, M.; Aspiotis, R.; Cauchon, E.; Chen, A.; Dubé, D.; Falgueyret, J.-P.; Fournier, P.-A.; Gallant, M.; Grimm, E.; Han, Y.; Juteau, H.; Liu, S.; Mellon, C.; Ramtohul, Y.; Simard, D.; St-Jacques, R.; Tsui, G. C. Bioorg. Med. Chem. Lett. 2012, 22, 1953-1957.
11. Synthesis of Unsymmetrical Polysubstituted Pyridines from β-Sulfonylvinylamines via 1-Aza-Allyl Anion Intermediates. Lau, C.; Tsui, G. C.; Lautens, M. Synthesis 2011, 3908-3914.
10. Rhodium(I)-Catalyzed Addition of Arylboronic Acids to (Benzyl-/Arylsulfonyl)acetonitriles: Efficient Synthesis of (Z)-β-Sulfonylvinylamines and β-Keto Sulfones. Tsui, G. C.; Glenadel, Q.; Lau, C.; Lautens, M.Org. Lett. 2011, 13, 208-211.
9. Linear-Selective Rhodium(I)-Catalyzed Addition of Arylboronic Acids to Allyl Sulfones. Tsui, G. C.; Lautens, M. Angew. Chem. Int. Ed. 2010, 49, 8938-8941. (highlighted in Synfacts)
8. Regioselective Rhodium(I)-Catalyzed Hydroarylation of Protected Allylic Amines with Arylboronic Acids.Tsui, G. C.; Menard, F.; Lautens, M. Org. Lett. 2010, 12, 2456-2459. (highlighted in Synfacts)
7. Procedure: Synthesis of Chiral Pyridine Bis(Oxazoline) Ligands for Nickel-Catalyzed Asymmetric Negishi Cross-Couplings of Secondary Allylic Chlorides with Alkylzincs: 2,6-Bis[(4R)-4,5-Dihydro-4-(2-Phenylethyl)-2-Oxazolyl]-Pyridine. Submitted by Lou, S.; Fu, G. C. Checked by Tsui, G. C.; Chai, D. I.; Lautens, M. Org.Synth. 2010, 87, 310-316.
6. Procedure: Nickel-Catalyzed Asymmetric Negishi Cross-Couplings of Racemic Secondary Allylic Chlorides with Alkylzincs: (S,E)-Ethyl 6-(1,3-Dioxolan-2-yl)-4-Methylhex-2-enoate. Submitted by Lou, S.; Fu, G. C. Checked by Chai, D. I.; Tsui, G. C.; Lautens, M. Org. Synth. 2010, 87, 317-329.
5. Patent: Invention relates to biaryl piperidine-based renin inhibitor compounds, and their use in treating cardiovascular events and renal insufficiency (Merck & Co.). WO2010114978 (A1). Publication date: 2010-10-07.
4. Synthesis of anti-2,7-Disubstituted Norbornadienes. Tsui, G. C.; Le Marquand, P.; Allen, A.; Tam, W.Synthesis, 2009, 609-619.
3. Iron-Catalyzed Cross-Coupling Reactions between a Bicyclic Alkenyl Triflate and Grignard Reagents. Le Marquand, P.; Tsui, G. C.; Whitney, J. C. C.; Tam, W. J. Org. Chem. 2008, 73, 7829-7832.
2. Palladium-Catalyzed Suzuki Couplings of 2,3-Dibromonorbornadiene: Synthesis of Symmetrical and Unsymmetrical Aryl-Substituted Norbornadienes. Yoo, W.-J.; Tsui, G. C.; Tam, W. Eur. J. Org. Chem. 2005, 1044-1051.
1. Ruthenium-Catalyzed [2+2] Cycloadditions Between Bicyclic Alkenes and Alkynyl Halides. Villeneuve, K.; Riddell, N.; Jordan, R. W.; Tsui, G. C.; Tam, W. Org. Lett. 2004, 6, 4543 – 4546.